Vector Systems
標準プラスミドCRISPRベクター
概要
CRISPR/Cas9ベクターは新しく開発されたゲノム編集ツールのひとつで、素早く効率的にゲノムの標的DNA配列に変異を導入できます(同様にZFNやTALENなどもよく利用されます)。
Cas9はRNA誘導性DNAヌクレアーゼの一種で、プラスミドやバクテリオファージなど外来遺伝子の侵入を防ぐ原生生物の自然免疫システムの一部です。Cas9は18‐22ntの標的配列と配列特異的な相互作用をするガイドRNA(gRNA)と複合体を形成します。gRNAの標的配列へのハイブリダイゼーションによってCas9はゲノムの標的配列を切断します。
CRISPRによる遺伝子ターゲティングには細胞内で標的配列特異的なgRNAとCas9が共発現している必要があります。Cas9とgRNAをひとつのベクター(all-in-oneベクター)から発現させるか、Cas9とgRNAをそれぞれ独立したベクターから発現させることによって共発現が可能になります。all-in-oneベクターを使うメリットは、CRISPRゲノム編集に必要なすべてのコンポーネントをひとつのベクターのみで供給でき、技術的に簡便である点です。独立したgRNAベクターとCas9ベクターの同時導入を試みても、両ベクターがすべての細胞に同時に導入される確率は大変低くなります。別の方法として、Cas9を安定して大量に発現する細胞もしくは生体にgRNAを発現させる方法がありますが、かなりの時間と労力が必要になります。当社のall-in-oneベクターはCas9とgRNAを標準プラスミドベクターから発現できるため、上記の困難を回避できます。
当社の標準プラスミドCRISPRベクターは従来の遺伝子導入法を使用して高い効率で哺乳類動物細胞にCas9とgRNAを導入できます。近年はレンチウイルスベクター、アデノウイルスベクター、AAVベクターやPiggyBac等の優れた遺伝子導入法が開発されていますが、従来通りの方法が技術的に容易であることと多くの細胞タイプにおいて十分な効率が得られるために、現在でも多くのラボで利用されています。標準プラスミドベクターによる遺伝子導入は一過性であり非常に少数の細胞(大抵は1%以下)でのみプラスミドのゲノムへの挿入が起こります。
標準プラスミドCRISPRベクターは実験目的に応じてシングルgRNA発現用とデュアルgRNA発現用のどちらかを選択できます。シングルgRNA発現ベクターはgRNA発現用のヒトU6プロモーターをひとつ、デュアルgRNA発現ベクターはふたつ持ちます。シングルgRNA発現ベクターは遺伝子ノックアウトなどの従来のCRISPRゲノム編集用途に利用され、デュアルgRNA発現ベクターは対となるゲノムサイトを同時に標的とする必要がある用途に利用されます。使用用途として次のようなケースがあります。1)標的サイトに対してペアとなるgRNAとhCas9ニッケース変異体(hCas9-D10A)を使用して、二本鎖DNAのそれぞれの鎖にニック(一本鎖切断)を入れてDSB(double strand break:二本鎖切断)を起こしてゲノム編集を実行する。2)対となるgRNAペアを利用して2ヵ所のDSBを作り出し、そのあいだの配列の欠失を作り出す。3)2つのgRNAによる2つの遺伝子の同時編集。
当社の標準プラスミドCRISPRベクターにはふたつの改良型Cas9(hCas9とhCas9-D10A)を利用できます。ヒトのコドン使用頻度に最適化されたhCas9は標的サイトで効率的にDSB(二本鎖切断)を作り出し、ニッケース変異体であるhCas9-D10Aは一本鎖ニックを標的サイトに作り出します。hCas9-D10Aが標的サイトのそれぞれのDNA鎖に対応するgRNAペアと共に使用されると、hCas9-D10Aは両DNA鎖それぞれに一本鎖ニックを入れることによって標的サイトにDSBを作り出します。この方法はDSBの形成にふたつのgRNAが標的配列を認識することが必要なため、シングルgRNAを利用する方法よりもオフターゲット効果の可能性を低下させ、標的配列への特異性を高めることができます。
細胞内での非相同性末端結合(nonhomologous end-joining pathway:NHEJ)によるDSBの修復は小規模な配列の欠損(稀に挿入および塩基置換)を引き起こします。これらの変異が遺伝子のタンパク質翻訳領域で発生すると(欠損、挿入が原因となるフレームシフトによって)、遺伝子機能の破壊につながります。効率は低くなりますが、CRISPR/Cas9ベクターと外来性ドナーDNAテンプレートを同時に導入することによって相同組み換え修復(homology-directed repair:HDR)によるDSBの修復も可能です。HDRによって標的となるゲノムDNA配列はDNAテンプレート配列に組み換えられるため、点変異などを導入可能です。ニックの入ったゲノムDNAには頻繁にHDRが起こるので、hCas9-D10AとDNAテンプレートを共導入すれば組み換えによる変異の導入が可能になります。
CRISPR/Cas9システムを利用すれば、ほとんどのDNA配列を標的とすることができます。しかし、gRNA標的配列の3’末端直後には必ずPAMとよばれる“NGG“配列(場合によっては”NAG”)が必要になります
当ベクターシステムに関する詳細な情報ついては下記の論文を参照してください
| References | Topic |
|---|---|
| Science 339:819-23 (2013) | Description of genome editing using the CRISPR/Cas9 system |
| Cell. 154:1380–9 (2013) | Use of Cas9 D10A double nicking for increased specificity |
| Nat. Biotech. 31:827–832 (2013) | Specificity of RNA-guided Cas9 nucleases |
特長
当社の標準プラスミドCRISPRベクターはE.coliでの高コピー数複製と高い遺伝子導入効率を実現します。当ベクターで形質転換された細胞はマーカー遺伝子によって薬剤選択もしくは可視化できます。標準プラスミドCRISPRベクターはひとつのプラスミドベクターからCas9とgRNAを共発現できるように設計されています。また、シングルgRNAもしくはデュアルgRNAを発現できるベクターを実験目的によって選択できます。
実験による検証
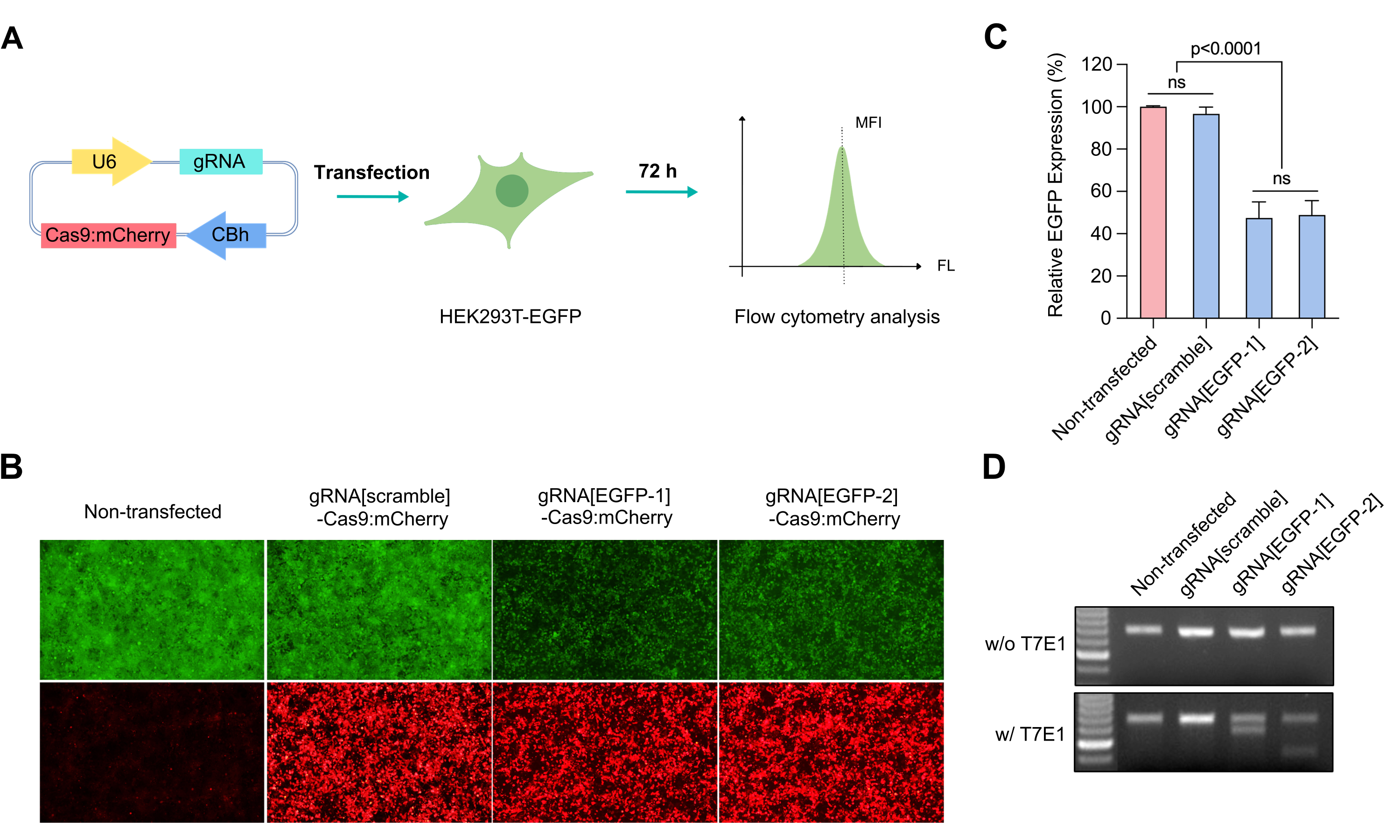
図1.all-in-one CRISPRシステムによる遺伝子編集の例。(A)EGFPを安定発現するHEK293T細胞(HEK293T-EGFP)にEGFPに対するgRNAまたはスクランブルgRNAをCas9-mCherryプラスミドと共トランスフェクションした。トランスフェクション後72時間にEGFPの発現をフローサイトメトリーにて解析した。MFIは平均蛍光強度(mean fluorescenceintensity)を示す。(B)EGFPおよびmCherryを蛍光顕微鏡下で観察した(倍率100Ⅹ)。(C)相対的なEGFPの発現レベルをall-in-oneCRISPRベクターのトランスフェ クションの有無で比較した。EGFP発現量の比は[MFI(all-in-oneCRISPRinHEK293T-EGFP) -MFI(HEK293T)]/[MFI(HEK293T-EGFP)–MFI(HEK293T)]にて計算した。Mean±SD,ns P>0.05,Tukey法によるANOVA。(D)ゲノムDNAからEGFPgRNAのターゲット領域をPCR によって増幅した。ゲノム編集はT7E1アッセイで確認した。
メリット
簡便さ: CRISPR/Cas9はgRNAと標的ゲノムサイトの配列の相同性によって機能するために、設計が簡単で分かりやすくなっています。当社の標準プラスミドCRISPRベクターは哺乳類細胞にCas9とgRNAを同時に導入できるように設計されています。これによってCRISPR遺伝子編集に必要なすべてのコンポーネントを一度に導入できるので、Cas9とgRNAを独立したベクターで導入するよりも簡便で時間を節約できます。
一過的発現:プラスミドCRISPRベクターは一過性かつ強力なCas9とgRNAの発現を実現します。ゲノム編集が完了した後は薬剤選択をやめることでプラスミドおよびCas9とgRNAは時間経過とともに細胞から取り除かれます。
デメリット
オフターゲット活性:CRISPR/Cas9システムはある程度のオフターゲット活性があります。一般的にはTALENシステムの方が低いオフターゲット活性を持つといわれています。しかしながら、標的のゲノムサイトに対してhCas9-D10AニッケースとgRNAを2つ使用して2本鎖DNAのそれぞれの鎖に対してニックをいれ、DSBを作り出す方法ならばオフターゲット活性を大幅に減少させることができます。
PAMが必要:CRISPR/Cas9システムはgRNA標的配列の3’末端のすぐ隣にPAMと呼ばれる配列が必要です。
基本コンポーネント
シングルgRNA 標準プラスミドCRISPRベクター
U6 Promoter: ヒトU6 snRNAのプロモーター。small RNAの転写をするRNAポリメラーゼIIIによってgRNAを高レベルで発現する。
gRNA: Streptococcus pyogenes由来のCas9用のgRNA。
Terminator: gRNAの転写を停止する。
CBh promoter: チキン β‐アクチンプロモーター。下流のCas9ヌクレアーゼを発現する。
Cas protein: Cas9ヌクレアーゼ(実験に応じてCas9もしくはhCas9-D10Aを選択)。
BGH pA: ウシ成長因子ポリアデニレーションシグナル。上流ORFの転写を停止する。
CMV promoter: ヒトサイトメガロウイルス(CMV)プロモーター。下流のマーカー遺伝子を遍在的に発現する。
SV40 late pA: SV40(Simian virus 40)のlateポリアデニレーションシグナル。上流マーカー遺伝子の転写を停止する。
Ampicillin: アンピシリン耐性遺伝子。 E.coliへのアンピシリン耐性によるプラスミドの維持を可能にする。
pUC ori: pUC複製起点。E.coliでプラスミドを高コピーで維持する。
デュアルgRNA 標準プラスミドCRISPRベクター
U6 Promoter: ヒトU6 snRNAのプロモーター。small RNAの転写をするRNAポリメラーゼIIIによってgRNAを高レベルで発現する。
gRNA #1: Streptococcus pyogenes由来のCas9用のgRNA#1。
gRNA #2: Streptococcus pyogenes由来のCas9用のgRNA#2。
Terminator: gRNAの転写を停止する。
CBh promoter: チキン β‐アクチンプロモーター。下流のCas9ヌクレアーゼを発現する。
Cas protein: Cas9ヌクレアーゼ(実験に応じてCas9もしくはhCas9-D10Aを選択)。
BGH pA: ウシ成長因子ポリアデニレーションシグナル。上流ORFの転写を停止する。
CMV promoter: ヒトサイトメガロウイルス(CMV)プロモーター。下流のマーカー遺伝子を遍在的に発現する。
SV40 late pA: SV40(Simian virus 40)のlateポリアデニレーションシグナル。上流マーカー遺伝子の転写を停止する。
Ampicillin: アンピシリン耐性遺伝子。E.coliへのアンピシリン耐性によるプラスミドの維持を可能にする。
pUC ori: pUC複製起点。E.coliでプラスミドを高コピーで維持する。



