MMLVレトロウイルス作製サービス
Moloney Murine Leukemia Virus (MoMoLV or MMLV)レトロウイルスベクターは、多様な動物種の増殖細胞に効率よく安定的に遺伝子導入を実施できます。特に近年、iPS細胞に遺伝子導入を行う際のウイルスベクターとして再認識されています。
VectorBuilderは独自に開発した技術と試薬を用い、MMLVのタイター、純度、生存率、均一性に優れたMMLVパッケージングプロトコールを開発しました。 VectorBuilderのMMLVベクター構築とウイルスパッケージングサービスに満足された世界中のユーザーに何度もご注文を頂いています。
ご提供しているMMLVレトロウイルスの種類
- VSV-Gシュードタイプ MMLVレトロウイルス
MMLVレトロウイルス受託作製サービスの詳細
価格と作業日数 プライスマッチ
| スケール | 推奨使用系 | タイター | 容量 | 価格(税別、送料別) | 作業日数 |
|---|---|---|---|---|---|
| パイロット | 培養細胞 | >107 TU/ml | 250 ul (10x25 ul) | 70,000円 | 6-12 日 |
| 中容量 | 1 ml (10x100 ul) | 101,000円 | |||
| 大容量 | >108 TU/ml | 1 ml (10x100 ul) | 170,500円 | ||
| 超純粋大容量 | 培養細胞 & in vivo | >108 TU/ml | 1 ml (10x100 ul) | 263,500円 |
TU = トランスダクションユニット(Transduction units、または感染ユニット)
価格は予告なく変更される場合があります。
納品形態
| 培養細胞実験用 | 生体用超純粋グレード |
|---|---|
| カスタムMMLVレトロウイルス | カスタムMMLVレトロウイルス |
|
無料: 標準コントロールウイルス
|
追加購入(オプション):超純粋標準コントロールウイルス
|
| 無料: ポリブレン (5 mg/ml, 200 ul) | 無料: ポリブレン (5 mg/ml, 200 ul) |
| メール添付:カルタヘナ法26条1項情報公開書類、品質検査証明書(COA) | メール添付:カルタヘナ法26条1項情報公開書類、品質検査証明書(COA) |
既成標準コントロールウイルスについて
カスタムウイルスをご購入いただく場合、 弊社既製品のコントロールウイルスを無料(パイロット、中容量、大容量用)または割引特価(超純粋大容量用)でご提供しています。既製標準コントロールウイルスは、感染効率の最適化などの目的に使用していただけるコントロールウイルスです。例えば、カスタムウイルスが目的遺伝子の強制発現系であれば、コントロールはEGFPを強制発現するウイルスです。厳密なカスタムコントロールが必要な場合は別途デザインとお見積りをご依頼ください。コントロールウイルスの詳細を以下に示します:
| ベクターシステム | コントロールベクター名称 | vector ID |
|---|---|---|
| MMLV遺伝子発現システム | pMMLV[Exp]-EGFP/Puro | VB010000-9307ddn |
| 自己不活性型 MMLV遺伝子発現システム | pMMLV-SIN[Exp]-MSCV>EGFP/Puro | VB010000-9478ycv |
保存方法
MMLVレトロウイルスはHBSSバッファーの溶液でチューブに分注し、凍結したものをドライアイス梱包で出荷・納品します。お受け取り後は、-80°Cで長期間(6か月)は安定です。-20°Cでは短期間(1週間)の安定保存が可能です。貯蔵寿命(シェルフライフ)は約1年です。 タイターの大きな低下を避けるために、凍結融解を繰り返すことは避けてください。
技術的情報
MMLVレトロウイルス作製と品質検査
MMLVレトロウイルスの製造では、目的の遺伝子(GOI)を運ぶトランスファーベクター、VSV-Gをコードする弊社独自のエンベロープベクター、GagおよびPolをコードするパッケージングベクターの3種類をウイルス産生細胞(パッケージング細胞)にコトランスフェクトします。短期間のインキュベーション後、上清を収集し、遠心分離して細胞破片を除去し濾過します。その後、ウイルス粒子をPEGで濃縮します。超純粋MMLVレトロウイルス(in vivoグレード)の場合、ウイルス粒子をさらに精製し、ショ糖濃度遠心分離によって濃縮します。MMLVの機能的タイターは、qPCRをベースとした方法で測定します。
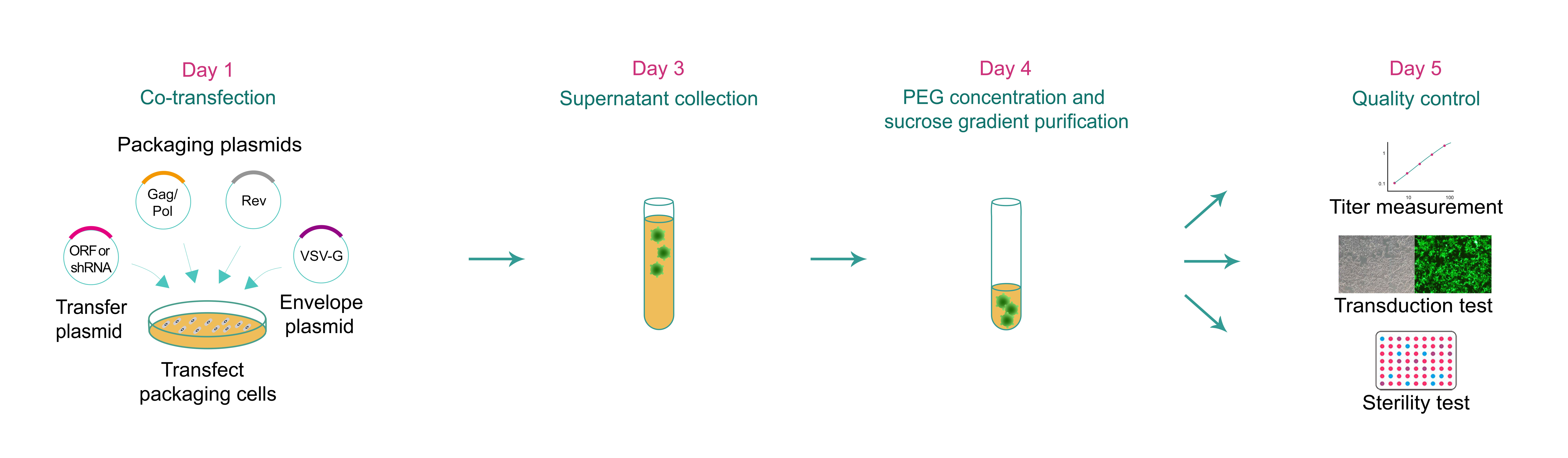
VectorBuilderによって作製された組換えMMLVレトロウイルスは、各ウイルスパッケージング毎に徹底した品質管理をしています。品質検査には、タイター測定、細菌および真菌の無菌試験、およびマイコプラズマ検出が含まれます。トランスファーベクターが蛍光タンパク質をコードしている場合、HEK293T細胞にウイルスを形質導入し、適切なインキュベーション後、該当する蛍光を蛍光顕微鏡下で検出します。さらに、超純粋MMLVの場合、定期的なサンプリングでエンドトキシンアッセイを実施してウイルス品質を管理しています。COAへエンドトキシンアッセイ結果の記載するためには追加費用が発生します。ご希望の追加QCにも対応可能です。
ご注文方法
ユーザー提供のMMLVウイルスプラスミドをパッケージに使用する場合は マテリアルサブミッションガイドライン に従ってマテリアルを準備し、弊社に発送してください。ユーザーからご提供されるマテリアルはVectorBuilderでお受け取り後、品質検査(QCチェック)に合格することを必須条件としています。QCチェックは、マテリアルごとに16,000円からの追加料金が発生する場合があります。さらにQCチェックに合格するまで、ご依頼の受託サービスが開始することができません。そのため、正しくマテリアルを準備、発送し、プロジェクトが遅延しないよう心がけてください。
リソース
技術的ドキュメント
ユーザーインストラクション 化学物質等安全データシート (MSDS) 品質検査証明書(COA)Q&A
| レンチウイルス | MMLV | アデノウイルス | AAV | |
|---|---|---|---|---|
| 組織指向性 | 広範 | 広範 | 数細胞種には不適 | ウイルスセロタイプに依る |
| 非増殖細胞への感染 | 感染する | 感染しない | 感染する | 感染する |
| 安定発現または一過性発現 | ゲノムへの挿入による安定発現 | ゲノムへの挿入による安定発現 | 一過的、エピソーマル | 一過的、エピソーマル |
| 得られる最高タイター | 高い | 中程度 | 高い | 非常に高い |
| プロモーターのカスタマイズ | 可 | 不可 | 可 | 可 |
| 主な使用系 | 培養細胞と in vivo | 培養細胞とin vivo | In vivo | In vivo |
| In vivoでの抗原性 | 低い | 低い | 高い | 非常に低い |
当社のタイター保証は弊社が構築したトランスファーベクターのうち、ウイルスにパッケージされている領域(5'MoMuLVLTRから3'MoMuLV LTRまで)がMMLVの搭載上限(8.3 kb)以下のものに適用されます。搭載上限の8Kbを超える場合でも、ベクターをウイルスにパッケージングすることは可能ですが、タイターが低下する可能性があります。以下のプラスミドベクターの場合、ウイルスタイターを保証することができません:
- 毒性遺伝子(例:アポトーシス誘導遺伝子)、パッケージング細胞またはウイルスの機能性を損なう遺伝子(例:細胞凝集を引き起こす膜タンパク質)など、パッケージングプロセスに悪影響を与える可能性のある配列を含むベクター、および内部欠失や組み換えなどのリアレンジメントまたは二次構造を取りやすい配列(例:反復的または非常にGCが豊富な配列);
- ユーザー提供のプラスミドベクターで、シークエンス非開示でパッケージングを依頼される場合、また非定型のMMLV機能要素(LTRなど)を含むプラスミドベクターの場合。弊社で最適化を図れないため、プラスミドパッケージング効率が不確実になる可能性があります。
推定作業日数は、弊社で受注後製造が開始し、QCに合格するまでの日数です。お客様から提供されたマテリアル(テンプレートDNAやウイルスベクターなど)の待ち時間、マテリアルのQCチェックに合格するまでのチェック日数、そして完成したウイルスを弊社製造拠点から出荷してお客様に納品するまでの移動日数は含まれません。





